The evidence behind Flow
See real patient outcomes and clinical effectiveness data.

Clinical
Results & Studies
Flow’s results are backed by extensive clinical research and real-world data from thousands of users. Here’s what our science says about our treatment approach.
Studies
In October 2024, the largest clinical trial into tDCS in the UK and US was published by the authoritative Nature Medicine.
Patients who used Flow for 10-weeks were about twice as likely to see their depression go into remission than those in a control group who performed the same procedure with the current switched off.
The placebo-controlled RCT is considered the gold standard method for evaluating efficacy and was carried out by Kings College London, the University of East London and UTHealth Houston.
Tackle more than just your low mood
66%* feel an improvement with their anxiety
Anxiety and depression can be intertwined. 2/3’s of our users felt an improvement of their anxiety after just 3 weeks of treating their depression with Flow.
Sleep better in less than 1 month
1/2 of users experience sleep improvements in 3 weeks
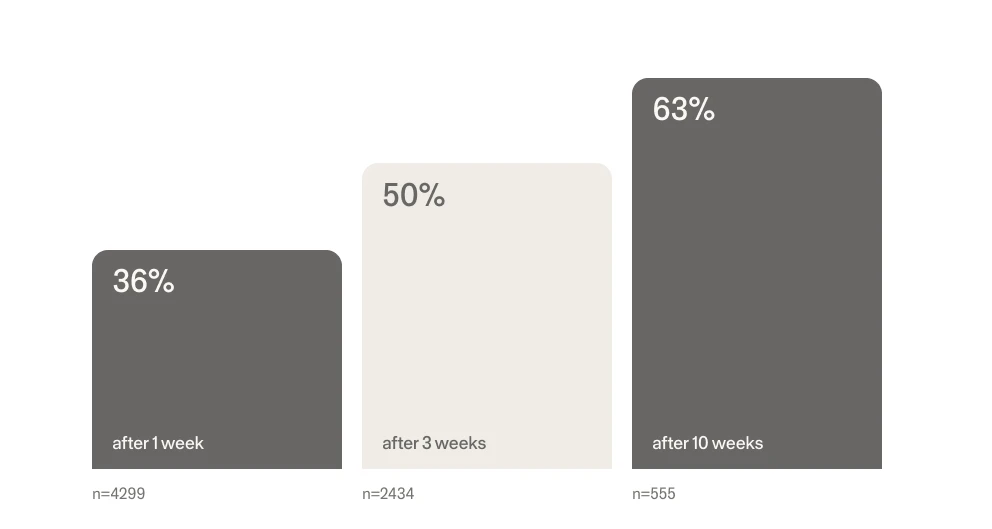
A retrospective-study with over 6,000 people showed that: 1/3 of users reported they no longer had insomnia after just 1 week and 2/3 reported being insomnia free by 10 weeks.
Reinforcing the importance and link between sleep and depression.
Depression-free by week 10
57% were in remission
In the largest clinical trial into tDCS across the UK and US, people using Flow were about twice as likely to see their depression go into remission than those in a control group who performed the same procedure with the current switched off (using a placebo device). 57% of those using Flow were depression-free after 10 weeks.
Feel better in 3 weeks
77% report improvement
We have over 55,000 people using the Flow headset across Europe.
Most people report positive results after 3 weeks; 77% see their depression score improve by at least 3 points. No one has reported a ‘serious’ side effect. Minor side effects include skin irritation and headaches.
Supported by
leading academics
"This is a potential first line treatment for depression. It can also be used for people whose depression hasn’t improved with antidepressant medication, for people who don’t like antidepressant medication, or who don’t want psychotherapy."

