The first and only FDA approved
tDCS at-home treatment for depression

FL-100
Flow Neuroscience
Flow FL-100 is an FDA-approved, home-based neuromodulation treatment for patients 18 and older with moderate to severe Major Depressive Disorder as a monotherapy or adjunct, without requiring prior medication failure.
Flow FL-100 is expected to be available in the US in the summer of 2026. Sign up to stay informed:
Potential amplification
when used as an adjunct†¹
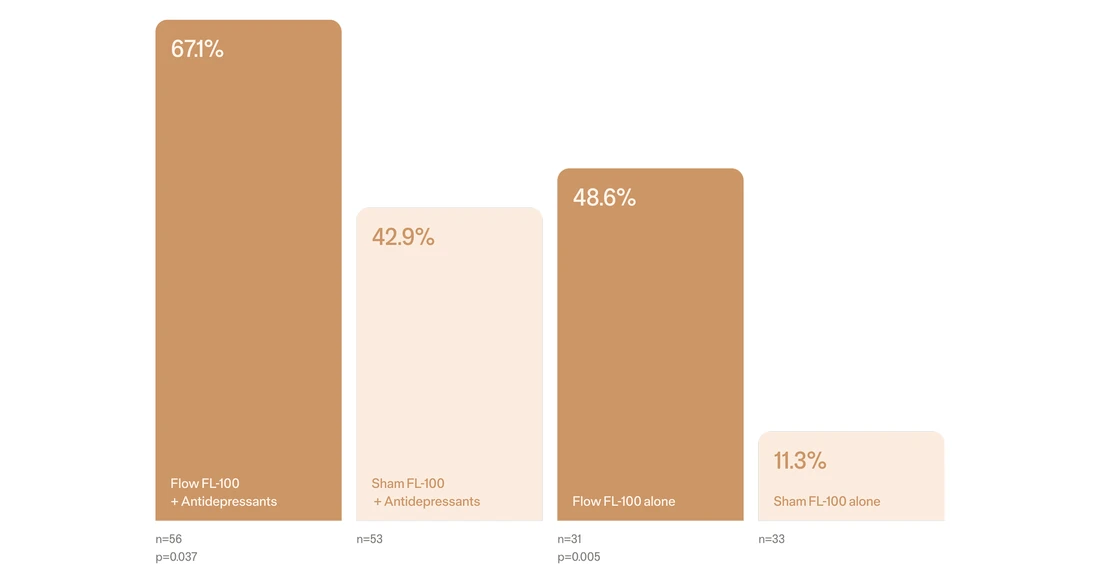
Results at 10 weeks from a multisite, double-blind, placebo-controlled randomised superiority trial demonstrate that Flow FL-100 is effective when used both as a standalone treatment and as an adjunct to antidepressant therapy.
Remission rates when used as an adjunct and standalone at Week 10†1

Flow FL-100 has shown a favorable safety profile to date, with both clinical trial1 and real-world user data suggesting a low incidence of adverse reactions.
Based on real world evidence from >40,000 users, the self-reported incidence rate of side-effects is 4.5%2.
Addressing risk in real-world clinical settings ‡
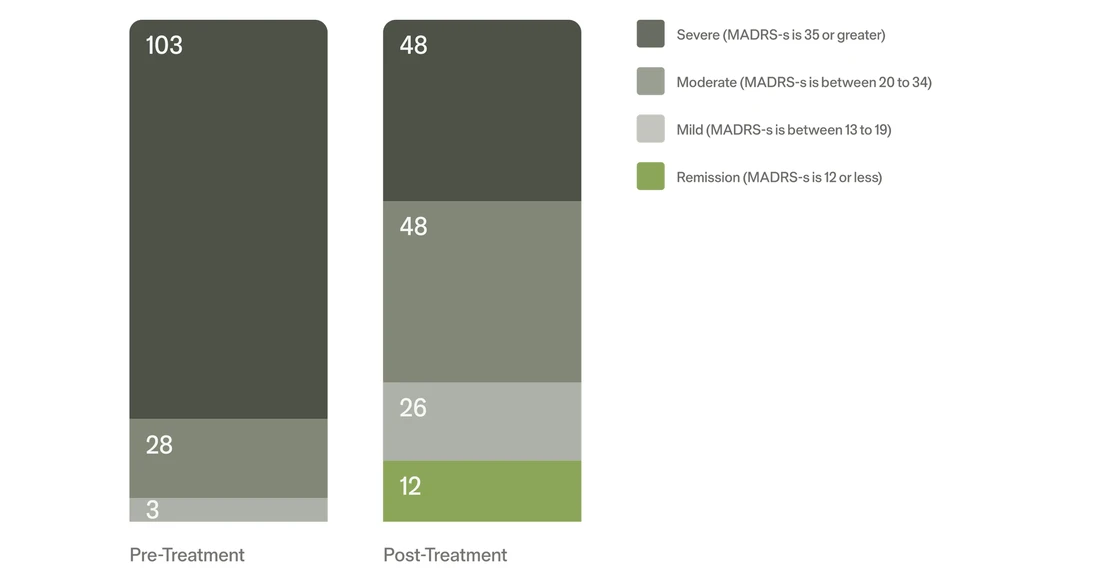
In a real-world crisis care evaluation3, Flow FL-100 use was associated with up to a 75% reduction in reported suicidal ideation and avoided psychiatric hospital admissions, suggesting potential value as a supportive intervention for patients with acute depressive symptoms.
Change in distribution of Depression Severity after 3 weeks3‡
References &
Footnote
*Remission is defined as a score of <10 on MADRS. MADRS remission rate was a secondary endpoint of the study. The primary endpoint of the study reviewed between-group mean difference in HDRS-17 scores and was –2.3 points in favour of the active group (p=0.012).
†Comparison of monotherapy and adjunctive use of Flow FL-100 was a sub-analysis evaluation included as table 10 and 11 in the Empower Study.
‡Findings from this crisis care evaluation are based on outcomes assessed after 3 and 6 weeks of treatment. The FDA-pivotal clinical trial evaluated its primary endpoint at 10 weeks; this difference in timeframe reflects the design and operational context of the crisis care service evaluation.
Please see more information about Flow FL-100, including the FDA-approved labelling, available here.