Both Transcranial Magnetic Stimulation (TMS) and transcranial Direct Current Stimulation (tDCS) are successfully used as depression treatments, but how exactly do they work and what’s the difference? Well, even though they target the same brain area, there are some important differences between the two brain stimulation techniques. For example:
- TMS is administered by health care professionals, whereas tDCS can be used at home.
- Most of the TMS research focuses on treatment “resistant” depression, whereas tDCS is primarily/usually recommended for mild to moderate depression.
This article aims to highlight these differences and similarities between TMS and tDCS. It will give you a detailed description of how TMS and tDCS work, their effectiveness in regards to depression treatment and an overview of the scientific history of each technique. In other words, this is your complete guide to TMS and tDCS for depression.
We’ll start by explaining Transcranial Magnetic Stimulation, move on to transcranial Direct Current Stimulation and end with an overall comparison between the two. Enjoy!
What is Transcranial Magnetic Stimulation (TMS)?
Before diving deep into the history and efficacy of Transcranial Magnetic Stimulation, let’s find out what TMS is and how it works. This part can get a bit technical, but it should be worth the trouble if you’re interested in understanding TMS. Here it comes:
With TMS, electromagnetism is used to alter the activity in specific areas of the brain. This means that TMS researchers and clinicians can change the activity of brain cells by using a magnetic field. The magnetic field is about the same strength as the one used in an MRI scan and the procedure typically lasts for 30-60 minutes. No anesthesia or muscle relaxants are needed and the patient is awake through the whole procedure.
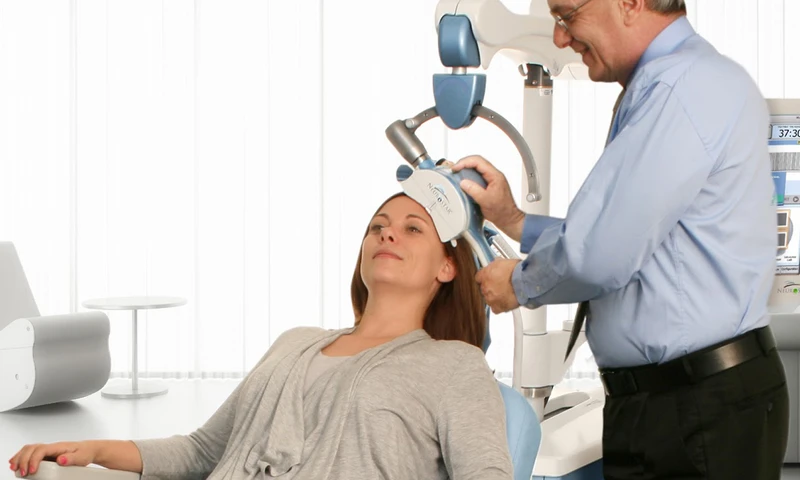
First, a health care professional places a coil against the patient’s scalp. In depression treatment, the coil is placed on the forehead to target a brain area in the front of the brain called the dorsolateral prefrontal cortex (DLPFC). This brain area plays an important role in depression (see the next section for more information about the DLPFC).

After that, electromagnetic pulses are passed through the coil. The magnetic pulses travel through the scalp and the skull and finally cause an electrical field in the DLPFC. The electrical field changes the activity of the brain cells and also affects neuronal networks connected to the area. So, why would the researchers and doctors want to do this? The next section will give you the answer.
Why does Transcranial Magnetic Stimulation work?
So far we’ve concluded that TMS changes the activity in the brain’s cortex and that the treatment helps against depressive symptoms, but why is that? What happens in the brain during TMS treatment? Well, TMS researchers, such as Janicak & Dokucu, explain the hypothesis behind TMS’s effectiveness. It reads something like this:
- Brain images of depressed patients show that depression is associated with lowered activity in an area in the front of the brain, called the DLPFC.
- When the activity in the DLPFC is unbalanced, it can lead to depressive behaviours, such as appetite changes, sleep disturbance and low energy levels.
- TMS treatment increases blood flow and metabolism in the DLPFC, which restores brain activity and decreases the depressive symptoms
Later on, you can study the scientific evidence for TMS treatment with the help of a TMS timeline, but first, let’s have a look at the difference between TMS and rTMS.
What is the difference between TMS and RTMS?
If you have searched the web for TMS, you may have noticed that the treatment is sometimes called Transcranial Magnetic Stimulation (TMS) and sometimes referred to as repetitive Transcranial Magnetic Stimulation (rTMS).
TMS and rTMS use the same technique and the same technical equipment. The only difference is that TMS creates a steady electromagnetic pulse, whereas rTMS creates several, repeated pulses. When used clinically, for example in depression treatment, rTMS usually involves several thousand pulses over a period of 30 to 60 minutes.
In general, TMS is used in experiments to explore the brain’s different functions and rTMS is used in the treatment of depression when clinicians want the changes in brain activity to last for a long period of time. So, when you read about TMS as a treatment for medical disorders, the procedure used is probably rTMS.
What about the scientific evidence for TMS?
To help you get an overview of the scientific evidence supporting the use of Transcranial Magnetic Stimulation for treating depression, we’ve created a timeline including the most important discoveries in TMS’s history. Take a look!
1985: The first TMS machine was invented
Transcranial Magnetic Stimulation (TMS) was originally invented by researcher and engineer Dr. Anthony T. Barker and his colleagues in 1985. Previous research had shown that pulses from magnetic fields could stimulate nerves in the body. Dr. Barker’s team expanded on this idea and invented a machine that could stimulate the cerebral cortex of the human brain, using electromagnets. The stimulation from the magnets caused less discomfort and disruptions than stimulation from the electric shocks used at the time.

1995: TMS helped patients when medication couldn’t
The first clinical research trial, exploring the effects of Transcranial Magnetic Stimulation for depression, was conducted. One of the most interesting things about this study was that it focused on six patients who were all “treatment-resistant”, meaning they had been given different types of antidepressant medications without effect. When given TMS, their scores on the Hamilton Rating Scale for Depression significantly decreased (lower scores mean less depression). The researchers concluded that:
“Daily left prefrontal rTMS appears to be safe, well tolerated and may alleviate depression”.
George et. al, 1995
1996-2014: High quality research proved the effectiveness of TMS for depression
Hundreds of research teams explored the effects of Transcranial Magnetic Stimulation for various types of illnesses, such as Major Depressive Disorder, Obsessive Compulsive Disorder, Schizophrenia and migraine. Since the first clinical study in 1995, over 35 randomized, sham-controlled trials have shown that TMS is an effective and safe treatment for depression. Read more about the scientific support for TMS in Janicak’s & Carpenter’s review of the evidence or Carpenter and colleagues’ multisite study.
2008: TMS became FDA approved
The first type of Transcranial Magnetic Stimulation was approved by the Food and Drug Administration (FDA) as a treatment for depression.
2010: The largest clinical TMS trial was published
Researchers conducted the largest clinical study on TMS and depression to date. They found that 14% of depressed patients received remission when given rTMS treatment, while only 5% of patients given sham treatment received the same results (sham treatment is like placebo, an inactive treatment that mimics the real procedure). After the first trial, all research participants, including those who were given sham treatment, were offered rTMS in a second trial. This time, almost 30% of depressed patients completely recovered from depression.
2013: The FDA approved “deep” TMS as depression treatment
Scientists explored the effects of a new type of Transcranial Magnetic Stimulation with custom made coils that could stimulate more deeply into the brain’s tissue. The technique was called “deep” TMS or dTMS and proved to be very effective in alleviating depressive symptoms for highly “treatment-resistant” patients with depression. In 2013, the FDA approved dTMS as a depression treatment for patients who are not helped by antidepressants or who can’t tolerate antidepressant medications. (Read more about the science behind dTMS for treatment-resistant depression here: Levkovitz et. al, 2015)
2015: Researchers concluded TMS is comparable to antidepressants
TMS researchers Philip G Janicak & Mehmet E Dokucu pointed out that Transcranial Magnetic Stimulation is as effective as antidepressants for treating depression. They also noted that, unlike studies of antidepressant medication, the TMS research has been conducted with the most difficult cases of depression:
“In summary, TMS is a promising, novel antidepressant treatment still relatively early in its development. Its efficacy and safety have improved significantly with continued research and clinical experience. The effect size for TMS antidepressant efficacy is at least comparable to those of antidepressant medications even though studies included only treatment-resistant or treatment-intolerant depressed patients.”
Get more information about TMS at the National Institute of Mental Health (NIMH).
What is transcranial Direct Current Stimulation (tDCS)?
Now, let’s switch focus to our second form of medication-free brain stimulation technique. If you suffer from depression, but don’t want to visit a health care facility, you have the option to treat depression at home using transcranial Direct Current Stimulation (tDCS). tDCS targets the same brain area as TMS, but uses electrical current instead of electromagnetic fields. (You’ll find a comparison between TMS and tDCS in the last section of this article.)

How does tDCS work?
tDCS is a form of brain stimulation that uses a low-strength electric current to help activate the brain cells in specific areas of the brain. tDCS has been used to treat depression for decades and should not be confused with electroconvulsive or ‘electroshock’ therapy (ECT). The tDCS current (0,5-2 mA) is 400 times weaker than the current used in electroconvulsive therapy and gives less severe and fewer side effects than antidepressant medication.
The tDCS current is delivered through two electrodes, placed on the scalp. When treating depression, the electrodes are placed high up on the forehead to target the brain area called DLPFC (for more information about the DLPFC, visit the next section). A brain stimulation session with tDCS is usually 30 minutes and can be administered at home with a tDCS headset.
tDCS headset (at-home): what it is and who it's for
A tDCS headset is a wearable device designed for home use that delivers the same brain stimulation therapy traditionally offered in clinical settings. The headset resembles a small headband with two electrodes that sit on the forehead, making it portable and convenient for regular use without clinic appointments.
Who benefits most from at-home tDCS:
- People with mild to moderate depression who prefer medication-free treatment
- Those unable to commit to regular clinic visits for TMS (30+ sessions)
- Patients seeking to supplement their existing depression treatment
- Anyone wanting accessible, evidence-based brain stimulation without prescriptions
Key features to look for in a tDCS headset:
- Medical certification: CE marking or FDA clearance ensures safety and efficacy standards
- Built-in safety features: Automatic shut-off, current monitoring, and placement guidance reduce risk
- App integration: Step-by-step instructions and progress tracking improve treatment consistency
- Clinical evidence: Published research demonstrating effectiveness specifically for depression
For a comprehensive breakdown of how tDCS headsets work, their effectiveness, and guidance on choosing the right device, see our complete guide to tDCS headsets.
Why does tDCS work?
The reason for tDCS’s effectiveness is basically the same as for Transcranial Magnetic Stimulation. As mentioned earlier, depression is associated with lowered activity in an area in the front of the brain, called the Dorsolateral Prefrontal Cortex (DLPFC), which can trigger depressive symptoms such as fatigue, sleeping problems, concentration difficulties and changes in appetite. The electrical current in tDCS stimulates brain activity in the DLPFC and thereby alleviates the depressive symptoms.
What about the scientific evidence for tDCS?
So far, we’ve concluded that tDCS uses electrical current to stimulate the brain’s DLPFC, which alleviates depressive symptoms. But how effective is tDCS for depression?
In 2017, Dr. Andre Brunoni, Assoc. Professor of Psychiatry at the University Hospital of Sao Paulo, and his colleagues explored the effectiveness of tDCS in a research study with 245 depressed participants. The researchers found that:
- 41% of the depressed participants discovered that 50% of their depressive symptoms had disappeared within 6 weeks of using tDCS.
- 24% of the participants completely overcame their depression within 6 weeks.
In 2019, doctoral researcher Julian Mutz and colleagues compared different forms of brain stimulation for depression. The researchers concluded that tDCS was an effective depression treatment and a less expensive option than TMS, ECT or psychotherapy.
“…we found tDCS to be efficacious across outcomes in both pairwise and network meta-analyses. Given that tDCS tends to be a less expensive treatment than transcranial magnetic stimulation, ECT, or psychotherapy, this finding is particularly relevant for policy makers who might consider tDCS as a clinical therapy outside the research setting.”
If you want to view a timeline of tDCS history, including the most important scientific discoveries since ancient times, click here.
Can you use tDCS at home?
As you’ve probably figured out by now, one big difference between Transcranial Magnetic Stimulation (TMS) and transcranial Direct Current Stimulation (tDCS) is that TMS is always administered by a professional at a health care clinic, whereas tDCS can be bought online and used at home. Flow Neuroscience has developed the first CE-marked and medically approved tDCS device for home use. It’s Bluetooth controlled and managed via the Flow Depression app. The Flow tDCS device uses the same technique as the devices in the research studies and you can get it here.
Quick guide to Flow's tDCS treatment:
- Order the Flow headset from the webshop.
- Download the free depression app.
- In a few days, your headset will be delivered. When it arrives, unpack your headset and start the app.
- The app will show you how to put your headset on and how to start the stimulation. Just follow the instructions.
- You’ll receive five 30-minute stimulation sessions per week, during the first three weeks. After that, there will be one or two sessions per week for as long as you like.
- Most people feel a reduction of their depressive symptoms after 3-4 weeks.
If you want to know more about how to use the Flow tDCS device, watch this video:
What’s the difference between TMS and tDCS?
In this section, you’ll find an overview of similarities and differences between Transcranial Magnetic Stimulation (TMS) and transcranial Direct Current Stimulation (tDCS).
Medical approvals
Both TMS and tDCS are medically approved treatments for depression.
- TMS: In 2008, TMS was approved by the Food and Drug Administration (FDA) as a depression treatment. TMS is used in hospitals in many countries around the world, including the USA, the UK and Sweden.
- tDCS: tDCS is approved for treating depression and chronic pain in the EU and the UK. The Flow tDCS device for depression is CE-marked and approved for medical use by the British Standards Institution (BSI).
At the clinic or at home
Neither TMS or tDCS require anesthesia or other medications. You can resume daily activities after the brain stimulation sessions.
- TMS: The treatment is administered by a health care professional at a clinic. Ask your doctor about TMS and where to get the treatment. The National Institute for Health and Care Excellence (NICE) has formulated some questions to ask your health care professional before receiving TMS.
- tDCS: You can complete the full treatment at home. The first medically approved tDCS device for home use is available to buy online.
Effectiveness
It’s difficult to make a fair comparison in this domain because most of the high quality research on TMS focuses on treatment resistant depression, whereas the research on tDCS focuses on all types of depression. However, brain stimulation research tells us that both TMS and tDCS are effective treatments. In 2019, Mutz and colleagues compared different forms of non-surgical brain stimulation and found both rTMS and tDCS to be effective treatments for Major Depressive Disorder (depression). 6750 participants were included in the study.
- TMS: In 2010, a research team at the University of South Carolina in Charleston found that as many as 30% of depressed patients could completely overcome depression with TMS treatment. If you suffer from stubborn, treatment resistant depression, TMS may be a helpful substitute for antidepressants
- tDCS: In 2017, a large research study showed that 41% of depressed patients had a 50% symptom reduction within 6 weeks and that 24% completely overcame their depression with tDCS. If you suffer from mild to moderate depression, tDCS may be the medication-free treatment for you (read more about the different levels of depression and take an online depression test here: Am I depressed?)
Treatment protocols
The exact number of stimulation sessions, as well as duration, varies for both treatments. Below, you’ll find a general overview of the treatment protocols.
- TMS: A TMS session is about 30-60 minutes long. A patient will usually have about 5 sessions per week and 20-30 sessions in total.
- tDCS: A tDCS session is about 30 minutes long. The recommended protocol includes 5 stimulation sessions per week during the first three weeks and, after that, one or two sessions per week. Most users notice a symptom reduction within 6 weeks.
Side effects
In comparison to antidepressant medications and electroconvulsive therapy (ECT), both TMS and tDCS have mild side effects. Side effects from tDCS are less severe than those from TMS.
- TMS: Side effects from Transcranial Magnetic Stimulation include mild to moderate headaches, muscle contractions during the procedure and a small risk of seizures. .
- tDCS: Side effects from transcranial Direct Current Stimulation include mild stinging sensations under the electrodes and mild headaches that pass after 30 minutes.
Hopefully, this article guided you through the history of TMS and tDCS with minimal effort. The important thing to remember is that both TMS and tDCS are effective treatments for depression and can be successfully used as alternatives to antidepressant medication.
Thank you for your attention!


